APA 7: TWs Editor & ChatGPT. (2023, November 9). Attaining Wellness: The Intricacies of Mitochondrial Care and the Harmony of Life and Death. PerEXP Teamworks. [News Link]
Mitochondria serve as the energy producers for all our bodily functions, supporting everything from physical movements to cognitive processes. These cellular powerhouses, which now reside within our cells, have ancestral roots in former free-living bacteria.
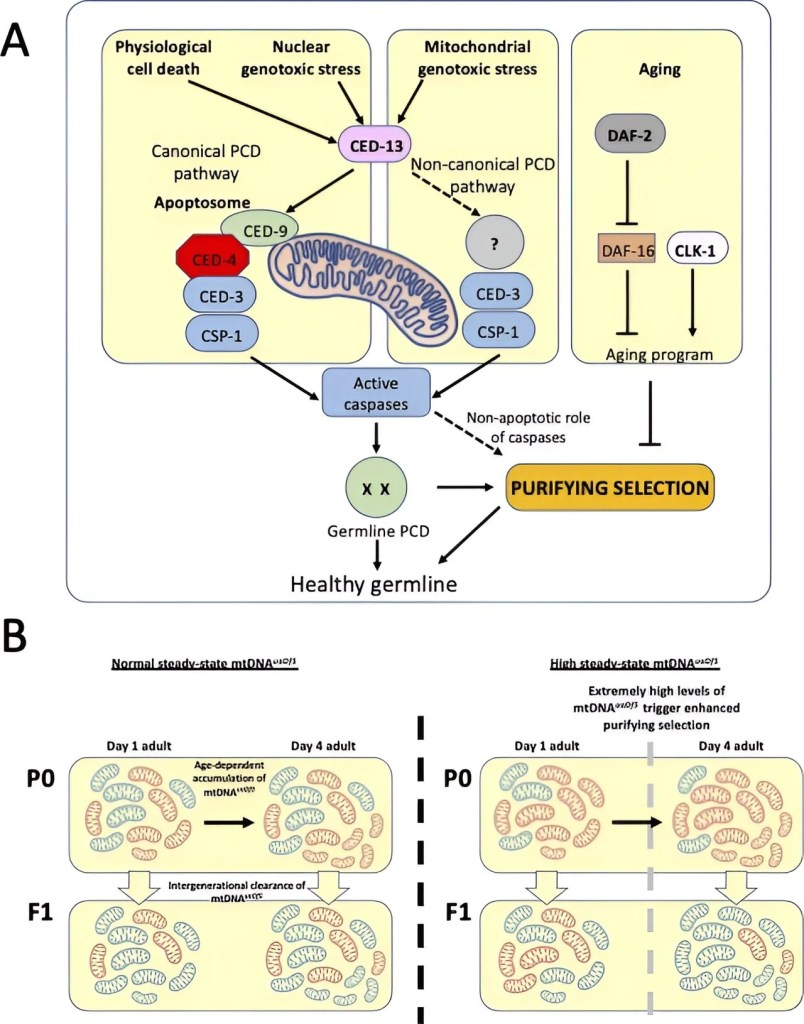
In the research conducted by Joel Rothman, a professor of molecular biology, it was observed that human beings represent a unique hybrid entity resulting from the fusion of two distinct evolutionary lineages. One lineage encompasses the mitochondria, originally autonomous bacteria, while the other comprises the cellular structures surrounding and hosting these mitochondria. This dual origin in our evolutionary history has led to the distribution of our DNA across two distinct compartments within every cell. The nucleus houses the majority of our genetic material, constituting most of our genome, while the mitochondria retain their own separate DNA, a relic of their ancient bacterial ancestry.
According to Joel Rothman, the accumulation of DNA damage in these cellular powerhouses, the mitochondria, plays a role in the age-related deterioration of our health. Their research findings shed light on a mechanism for eliminating malfunctioning mitochondria, leading to a rejuvenation of the cells.
The study, which was recently published in the journal eLife, demonstrates that the biological apparatus responsible for acting as a “cellular kill switch” to eliminate potentially harmful cells, such as those undergoing cancerous changes, also plays a role in the removal of flawed mitochondrial DNA.
Pradeep Joshi, a co-author of the publication and a senior scientist, highlighted the dual nature of mitochondria. On one hand, they are responsible for generating the energy that supports life. On the other hand, with each breath, mitochondria also produce reactive oxygen species, which are detrimental molecules capable of harming DNA and various cellular components.
Hence, as our lifespan extends, the cumulative damage intensifies. This damage, in turn, diminishes the efficiency of energy production by our mitochondria, thereby impacting our overall healthspan. Given that critical organs like the heart, muscles, and brain rely heavily on this energy supply, the aging process is inherently linked to conditions like heart failure, reduced muscle function, and cognitive decline. Pradeep Joshi suggests that the aging process can be viewed as a type of mitochondrial disorder. In his view, the removal of mitochondrial damage has the potential to enhance both healthspan and lifespan.
The research group identified a method to eliminate impaired mitochondria by employing a tiny nematode known as C. elegans, a creature that has made significant contributions to biomedicine, recognized in several instances through six Nobel prizes. The study revealed that the enzymes responsible for cell destruction also play a crucial role in eliminating damaged mitochondrial DNA. When these enzymes are lacking, malfunctioning mitochondria accumulate within the cells.
To the surprise of Rothman and his colleagues, they discovered that while certain proteins are shared, the overall mechanism for eliminating damaged mitochondria differs from the standard process used to clear excess cells. Pradeep Joshi noted that the cellular machinery responsible for cell death seems to be adapted for the purpose of clearing out dysfunctional mitochondria. In doing so, it contributes to the restoration of the health and function of these crucial cellular powerhouses.
In this study, it’s important to note that mitochondrial DNA, inherited solely from one’s mother, was a shared characteristic among humans and the animals under examination. The research uncovered a significant finding: as mothers age, the accumulation of faulty mitochondria within their cells increases. Joel Rothman emphasized the concerning consequence of this aging-related phenomenon, which is the transmission of these flawed mitochondria to their offspring.
The positive outcome from this research is that there is a potential solution to lessen the build-up and inheritance of flawed mitochondria. The scientists determined that a single genetic alteration, which slows down the aging process in animals and extends their lifespan, can alleviate these issues.
Joel Rothman, who also serves as the founding Director of the Center for Aging and Longevity at UCSB, observed that the deceleration of the aging process seems to lead to a reduced rate of accumulation of flawed mitochondria. This finding suggests that anti-aging interventions have the potential to yield healthier mitochondria.
These findings offer insights into potential approaches for eliminating compromised mitochondria and revitalizing cells, which could pave the path toward granting everyone additional years of vigorous and ailment-free living to relish.
Resources
- NEWSPAPER Fernandez, S. & University of California – Santa Barbara. (2023, November 8). Zen and the art of mitochondrial maintenance: The machinery of death makes a healthier life. Phys.org. [Phys.org]
- JOURNAL Flowers, S. E., Kothari, R., Cleuren, Y. N. T., Alcorn, M. R., Ewe, C. K., Alok, G., Fiallo, S. L., Joshi, P. M., & Rothman, J. H. (2023). Regulation of defective mitochondrial DNA accumulation and transmission in C. elegans by the programmed cell death and aging pathways. eLife, 12. [eLife]
















