Men and women experience pain differently, and until recently, the underlying reasons remained unclear. New research indicates that these differences might be partly due to variations in male and female nerve cells.
Researchers found that pain-sensing nerve cells from male and female animal tissues reacted differently to the same sensitizing substances. This discovery, reported on June 3 in Brain, suggests that at the cellular level, pain is generated differently between the sexes.
This finding has significant implications for the development of targeted pain treatments. According to Katherine Martucci, a neuroscientist specializing in chronic pain at Duke University School of Medicine, who was not involved in the study, the observed differences in the cells might pave the way for drugs tailored specifically for either female or male patients. These cellular differences confirm that there is no debate about the distinct mechanisms of pain in males and females.
Certain types of chronic and acute pain are more prevalent in one sex, but the reasons for this remain unclear. For example, around 50 million adults in the United States suffer from chronic pain conditions, many of which are more common in women. Similar disparities are observed in acute conditions.
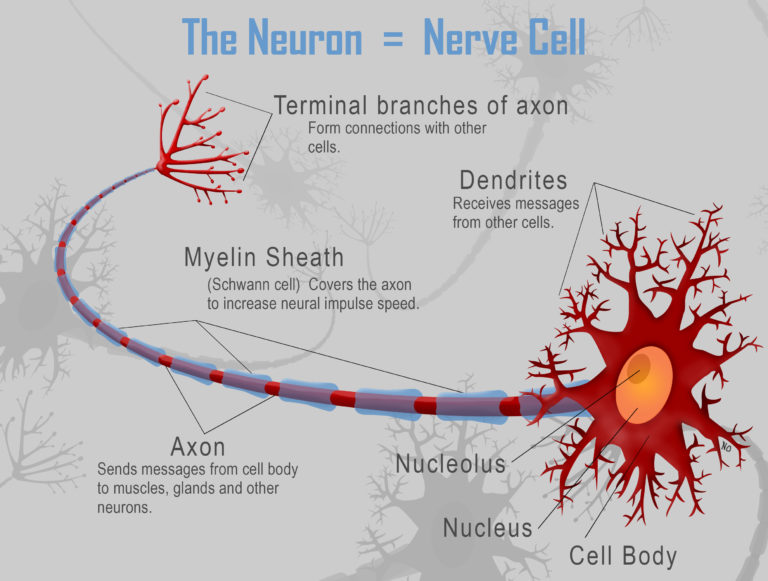
These differences led Frank Porreca, a pain researcher at the University of Arizona Health Sciences in Tucson, and his colleagues to investigate nerve cells known as nociceptors. These nociceptors function like alarm sensors for the body. Found in the skin, organs, and other parts of the body, these cells detect potentially harmful stimuli and send signals to the brain, which then interprets the information as pain. In certain cases, these nerve cells can become hypersensitive to external stimuli, perceiving even gentle sensations, such as a shirt brushing against sunburned skin, as pain.
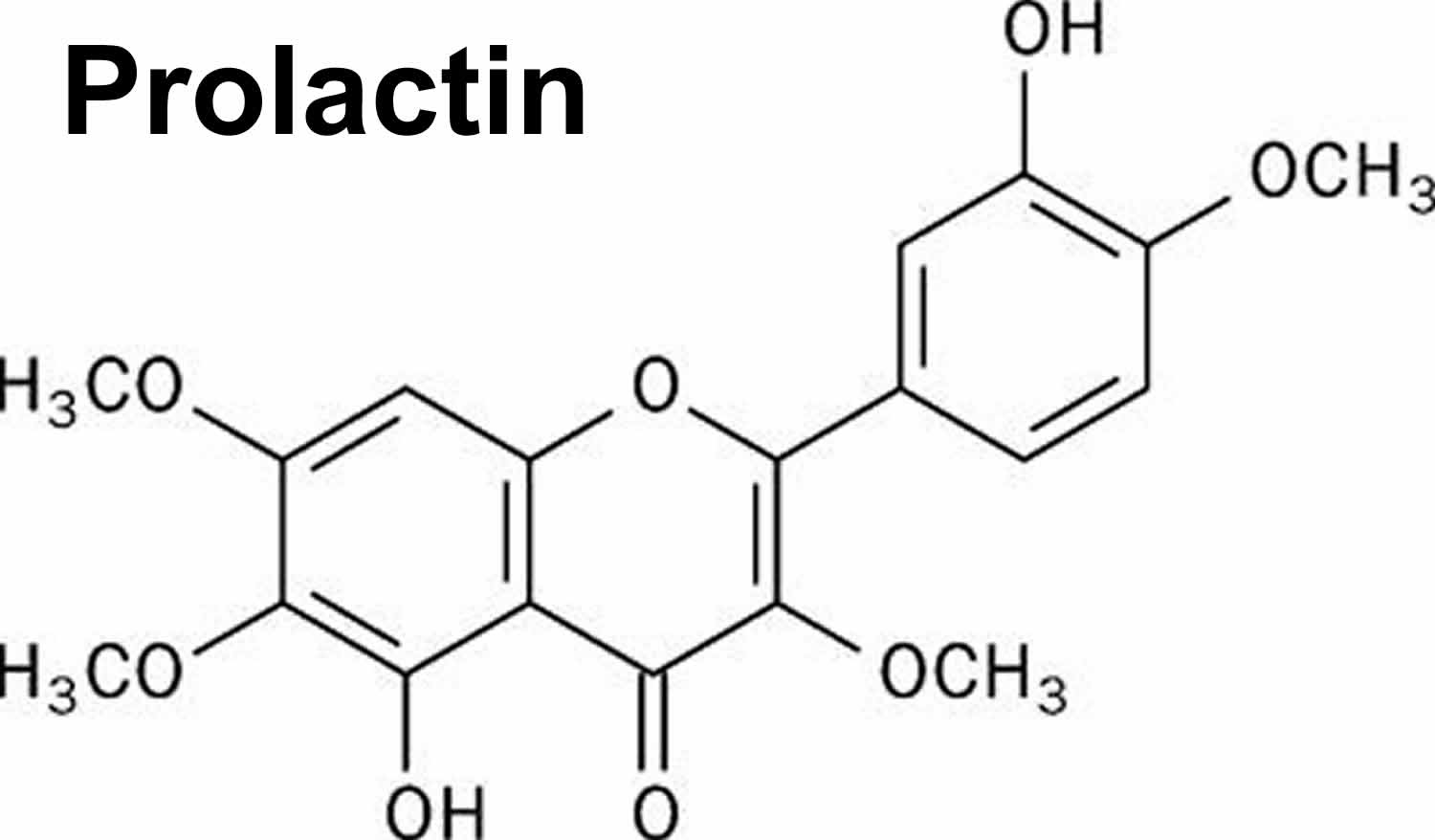
The researchers examined tissue from mice, monkeys, and humans to study the effects of two substances that can make nerve cells more sensitive to pain: a hormone called prolactin and a neurotransmitter named orexin B. When nerve cells encounter these substances, they become more responsive to stimuli, lowering the threshold at which they send electrical signals to the brain to indicate something is wrong. In all three species, exposure to prolactin increased the activity of nerve cells from females, while orexin B had a similar effect on those from males.
The research data reveal significant differences between men and women even at the initial stage of the pain pathway, according to Harrison Stratton, a neuroscientist at the University of Pittsburgh.
Given that the mechanisms producing pain differ between sexes, this suggests that pain (pathways) medications could be tailored specifically for males or females. For instance, treatments might involve blocking sensitivity to prolactin in females and to orexin B in males. These substances are already well-studied for their roles in other physiological processes like lactation and sleep. Existing U.S. Food and Drug Administration-approved orexin blockers, currently used for treating insomnia, could potentially be repurposed for managing pain. Furthermore, Porreca and his team previously identified a prolactin-blocking antibody, which holds promise for treating various conditions, from female-specific pain to infertility.
While findings from mouse studies often do not directly apply to humans, Katherine Martucci notes that the specific pain pathway investigated in this study appears to function similarly across mice, monkeys, and humans. This similarity suggests that future drugs targeting prolactin and orexin B could be effective in managing pain, pending further research and testing. This offers a clear direction for developing new pain treatments.
However, Richard Miller, a pharmacologist at Northwestern University Feinberg School of Medicine, points out that while the study highlights sex differences in pain mechanisms, pain itself is often influenced by a multitude of factors. This implies that other substances might also produce similar effects, raising questions about whether prolactin and orexin B are uniquely critical targets for pain management.
- ONLINE NEWS Yuan, C. (2024, June 20). Pain may take different pathways in men and women. Science News. [Science News]
- JOURNAL Stratton, H., Lee, G., Dolatyari, M., Ghetti, A., Cotta, T., Mitchell, S., Yue, X., Ibrahim, M., Dumaire, N., Salih, L., Moutal, A., François-Moutal, L., Martin, L., Navratilova, E., & Porreca, F. (2024). Nociceptors are functionally male or female: from mouse to monkey to man. Brain. [Brain]
- JOURNAL Maciuba, S., Bowden, G. D., Stratton, H. J., Wisniewski, K., Schteingart, C. D., Almagro, J. C., Valadon, P., Lowitz, J., Glaser, S. M., Lee, G., Dolatyari, M., Navratilova, E., Porreca, F., & Rivière, P. J. (2023). Discovery and characterization of prolactin neutralizing monoclonal antibodies for the treatment of female-prevalent pain disorders. MAbs, 15(1). [MAbs]
- BOOK CHAPTER Osborne, N. R., & Davis, K. D. (2022). Sex and gender differences in pain. In International Review of Neurobiology (pp. 277–307). Academic Press.
APA 7: TWs Editor. (2024, June 20). Pain Pathways: Yes, They’re Gender-Specific!. PerEXP Teamworks. [Online News Link]

















Gosh – how fascinating… I feel the need to go do some more research based on this!
Linda xx