APA 7: TWs Editor. (2023, November 25). The Biodistribution and Role of Polymer-DNA Origami Nanostructures in Living Systems. PerEXP Teamworks. [News Link]
Through the formation of electrostatic interactions with polyamine, the team achieved enhanced stability of the agents in vivo. Following subcutaneous injection, over 90% of the components retained their structural integrity for a duration of five days, highlighting the robustness of the formulation.
The results underscore the potential of polymer-DNA hybrid nanostructures as effective pharmacological entities, positioning them for integration into mainstream technologies. This includes their application as monoclonal antibodies to facilitate drug activity.

(b) Assessment of DNA origami nanostructure quality through gel electrophoresis. Lane 3, 6, 9 corresponds to post-assembly analysis, lanes 4, 7, 10 after PEG purification, and lanes 5, 8, 11 after PEG-polylysine addition. A 1 kb double-stranded DNA ladder serves as a reference, with specific bands (indicated in kb). Red arrows denote staple excess, green arrows signify well-folded nanostructures pre- and post-PEG purification, and black arrows indicate PEG5K-K10-coated purified nanostructures.
(c) Visualization of DNA origami nanostructures via transmission electron microscopy (TEM). Each structure is imaged before and after PEG-poly(lysine) coating, with scale bars set at 100 nm for reference.
(Scientific Reports)
The potential of DNA origami as a therapeutic tool
Numerous drugs, encompassing both small molecules and biologicals, often lack inherent mechanisms for targeted distribution and action within the body. This inherent limitation serves as a primary catalyst for adverse effects and constitutes a significant factor contributing to the compromised efficacy of many novel drugs undergoing clinical trials and those in clinical use.
Despite substantial efforts over the past decades to regulate drug activity, the currently approved drugs only scratch the surface of the vast therapeutic potential inherent in the mechanisms of drugs.
The challenge is evident in widely embraced and established pharmaceutical approaches, such as monoclonal antibodies. These drugs have ushered in groundbreaking therapies for conditions previously deemed almost untreatable, particularly in the realms of oncology, immunology, and inflammatory diseases. Another avenue, the Scaffolded DNA origami technique, emerges as a method to engineer DNA nanostructures, providing a means for precise spatial control and functionality at scales below a nanometer.
The innovation of DNA therapeutics: A new way to treat diseases
The distinctive characteristics make these entities well-suited for diverse research fields, positioning them as advanced agents for both therapeutic and diagnostic applications in the next generation. Various methods for functionalizing DNA origami offer a greater degree of functional complexity compared to monoclonal antibodies.
In the innovative approach introduced by Joseph and collaborators, the team orchestrated spatial control over drug activity by merging polymer-DNA origami hybrid nanoscale agents. These conceptualizations can be customized for multiple target proteins, presenting a versatile solution for a broad spectrum of therapeutic functionalities.
Within this study, Joseph and team outlined a methodology for delivering therapeutic drug components using polymer-DNA origami hybrid nanoscale compounds. Through standard kinetic and stability characterization studies of different DNA origami constructs in vivo, the researchers identified an optimal DNA nanostructure as a proof-of-principle for therapeutic applications, showcasing potent anti-inflammatory effects in both a mouse model and human Tumor Necrosis Factor alpha scenarios.
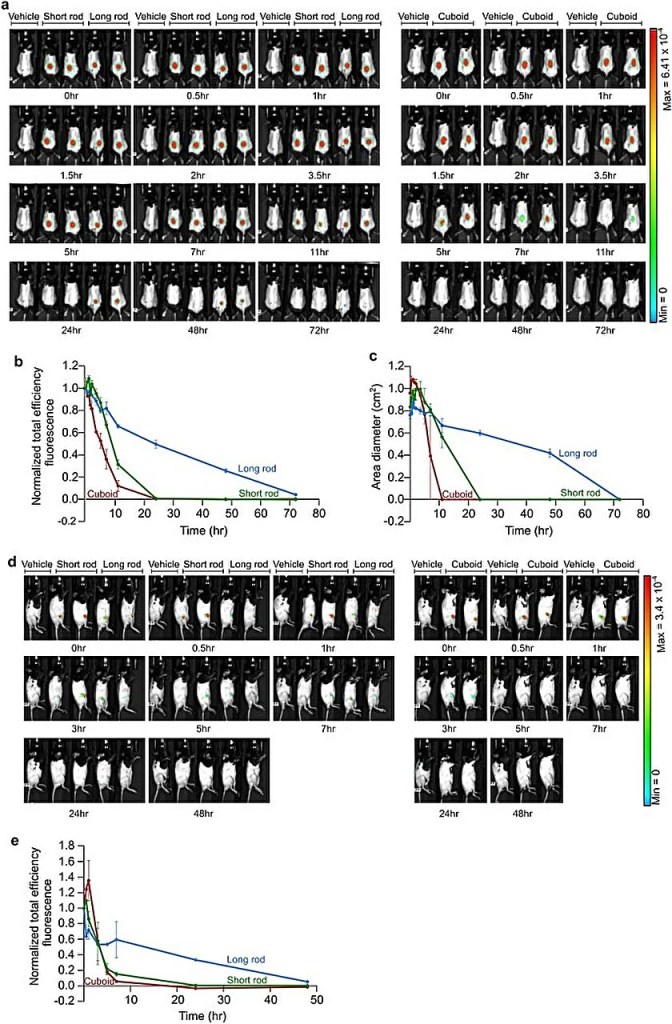
(b) Quantification of the total efficiency fluorescence observed in mouse images from (a). A consistent region of interest (ROI) was selected around the injection site for each mouse, and FRET fluorescent total efficiency for the specified DNA origami nanostructures was measured over different time points. Calculations were conducted according to the outlined “Methods” section. The presented data represents mean values ± SEM.
(c) Evaluation of the diffusion patterns of the indicated DNA origami nanostructures over time after subcutaneous injection into mice. Calculations were performed using mouse images from (a). Data is presented as mean values ± SEM.
(d) Live imaging analysis illustrating the dynamic biodistribution of specified DNA origami nanostructures over time following their injection into the knee joints of mice. False color heat maps are indicative of FRET levels.
(e) Quantification of total efficiency fluorescence derived from mouse images in (d). The same ROI around the injection site was chosen for each mouse, and FRET fluorescent total efficiency for the mentioned DNA origami nanostructures was measured at various time points. Calculations were carried out following the outlined “Methods.” The data presented represents mean values ± SEM. (Scientific Reports)
The explorations and discoveries
Initiating a proof-of-feasibility investigation, the research team opted for three distinct DNA origami nanostructures with comparable mass. Gel electrophoresis was employed for bulk quality assessment, and transmission electron microscopy captured the nanostructures’ morphology before and after coating with polyethylene glycosylate-polylysine. This coating, achieved through amine and phosphate interactions, aimed to augment DNA mass, enhance attachment to polyethylene glycosylate, and ensure the stability of the DNA origami structures.
Emphasizing the importance of in vivo stability for effective drug distribution, the team conducted live imaging studies on mice treated with subcutaneously administered polymer-coated nanostructures, targeting knee joints or delivered intraperitoneally.
While the long rod exhibited prolonged diffusion over time, a strategic balance between slower diffusion and heightened stability was achieved through subcutaneous administration. The team delved into the kinetics and in vivo stability of these observations, ultimately identifying the polymer-coated rod nanostructures with optimal polymer counts as efficacious candidates for subsequent druggability experiments.
DNA origami nanostructures and their therapeutic impact
Examining the reengineered long rod nanostructures designed to mimic human tumor necrosis factor alpha aptamers, the researchers uniformly incorporated them across the surface structures. Joseph and collaborators scrutinized the functionalization of these long rod DNA origami structures, employing agarose gel electrophoresis, transmission electron microscopy, and atomic force microscopy for detailed analysis.
To assess the stability of the constituents, the team subjected them to a 10-day exposure to human serum, meticulously tracking their structural integrity for subsequent biodistribution and in vivo studies.
Overview
Noah Joseph and his research team elucidate the in vivo kinetics of three distinct DNA origami nanostructures, each assuming different shapes but stabilized by the polyethylene glycol-polylysine polymer. Identifying the optimal candidate, the scientists further functionalized the long rod nanostructures by affixing human tumor necrosis factor alpha aptamers, specifically targeting the corresponding protein.
In detailing the therapeutic promise of these functionalized co-polymer DNA origami nanostructures, the research team underscores their capacity to navigate complex biological environments. Collectively, these findings underscore the pivotal role of DNA nanostructures as potent therapeutic agents, showcasing their potential in precision medicine and advancing the functionality of therapeutic interventions.
Resources
- ONLINE NEWS Jeewandara, T. & Phys.org. (2023, November 24). Examining the biodistribution and function of polymer-DNA origami nanostructures. Phys.org. [Phys.org]
- JOURNAL Joseph, N., Shapiro, A., Gillis, E., Barkey, S., Abu-Horowitz, A., Bachelet, I., & Mizrahi, B. (2023). Biodistribution and function of coupled polymer-DNA origami nanostructures. Scientific Reports, 13(1). [Scientific Reports]
















