The quotes in this article have been changed in a way that does not change the intended meaning. You can find the original quotes in the resources.
Yujie Wang, a co-author of a recent paper on this fuel and a doctoral student in chemical engineering at UCR, pointed out, “The typical fuels we use pose significant safety risks due to their flammability and evaporative nature, making them challenging to manage. Our fuel offers a simpler way to regulate flammability – it can be easily extinguished by simply removing the voltage.”
In a paper published in The Journal of the American Chemical Society, the researchers outline the process of developing this unique fuel. Further technical specifics are available in the patent they’ve submitted.
During combustion, it’s not the liquid fuel that actually burns, but the volatile molecules emanating from the liquid’s surface that ignite upon contact with oxygen and a flame. Halting the flame by eliminating the oxygen source is challenging, especially outside of controlled engine environments.
“When a match meets a pool of gasoline, it’s the gas vapor that catches fire. You can detect the volatility by its distinct odor,” explained Prithwish Biswas, first author of the paper and a UCR chemical engineering doctoral student. “By managing the vapor, we can determine if the fuel ignites.”
The foundation of this novel fuel is an ionic liquid, a type of liquefied salt. “It resembles the salt we sprinkle on our meals, like sodium chloride,” explained Wang. “The variant we employed in this endeavor has a lower melting point than table salt, exhibits low vapor pressure, and is organic in nature.”
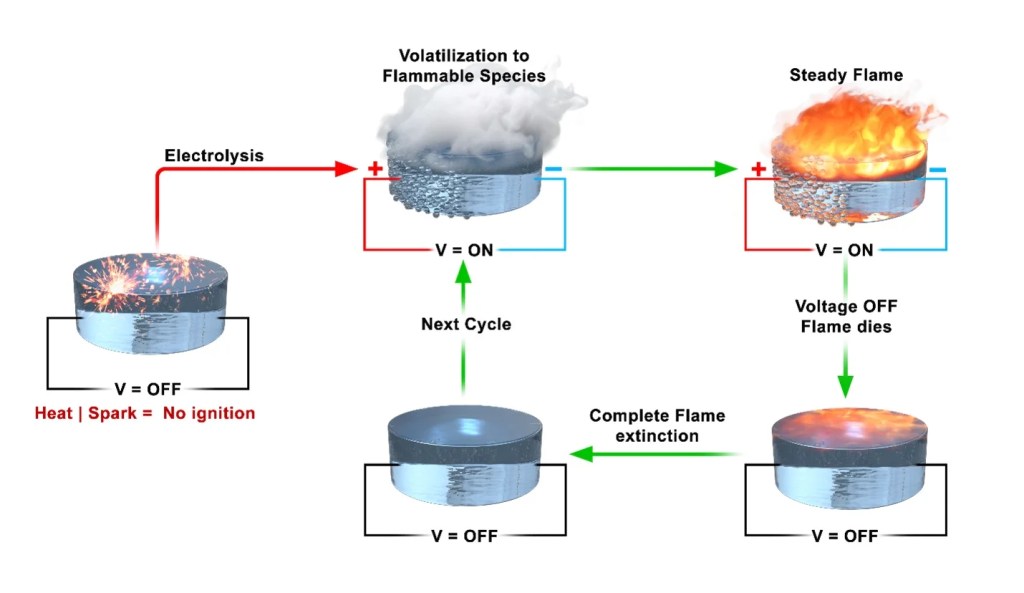
Inside their laboratory, the researchers tweaked the ionic liquid’s composition, substituting chlorine with perchlorate. Subsequently, they employed a common cigarette lighter to test if the resulting liquid would catch fire. “The temperature generated by a standard lighter is sufficiently high, and if ignition were likely, it would have occurred,” remarked Wang.
They then proceeded to test it further by applying an electric voltage followed by a lighter’s flame, which resulted in ignition. “When we cut off the current, the flame extinguished, and we could replicate this process repeatedly—applying voltage, observing smoke, igniting the smoke, and then deactivating it,” explained Wang. “This discovery excited us because it allowed us to create a system that could be rapidly initiated and halted.”
Increasing the voltage applied to the liquid led to the generation of larger flames with higher energy output. This characteristic could potentially serve as a control mechanism akin to a metering or throttling system within an engine.
“This method enables combustion measurement, and reducing the voltage serves as a safety feature, similar to a dead man’s switch, which automatically shuts down a machine if the operator becomes unable to do so,” explained Michael Zachariah, a distinguished professor of chemical engineering at UCR and one of the authors of the paper.
In theory, the ionic liquid fuel holds potential for use in various vehicle types. Nonetheless, several critical questions must be addressed before it can enter commercial use. Comprehensive engine tests across different vehicle types and a thorough evaluation of its efficiency are prerequisites for its potential commercialization.
A fascinating characteristic of the ionic liquid is its compatibility with traditional fuels when mixed. However, further investigation is necessary to determine the maximum blend ratio that maintains its non-flammable properties, as pointed out by Zachariah.
While there are areas avenues for further research on this liquid, the team is enthusiastic about their achievement of creating a fuel impervious to accidental or inadvertent ignition.
“It’s worth noting that this approach may incur higher production costs compared to the current methods of fuel manufacturing. These compounds aren’t typically mass-produced, but if they were, we could anticipate a reduction in costs,” explained Zachariah.
“How would its competitiveness stack up? That’s uncertain at this point. However, if prioritizing safety is crucial, that’s a significant facet of this development. Ensuring safety has inherent value that extends beyond mere financial considerations,” commented Zachariah.
Resources
- WEBSITE Bernstein, J. (2023, September 28). Scientists unveil fire-safe fuel. UC RIVERSIDEV | News. [UC RIVERSIDEV | News]
- JOURNAL Biswas, P., Wang, Y., Hagen, E., & Zachariah, M. R. (2023). Electrochemical modulation of the flammability of ionic liquid fuels. Journal of the American Chemical Society, 145(30), 16318–16323. [Journal of the American Chemical Society]
















